Do You Have to Start All Over Again if You Forgot to Zero Your Blank for Your Absorbances
1.2: Beer'southward Law
- Page ID
- 111324
What factors influence the absorbance that you lot would measure for a sample? Is each cistron straight or inversely proportional to the absorbance?
One factor that influences the absorbance of a sample is the concentration (c). The expectation would be that, as the concentration goes upwards, more than radiation is captivated and the absorbance goes up. Therefore, the absorbance is directly proportional to the concentration.
A 2d gene is the path length (b). The longer the path length, the more molecules there are in the path of the beam of radiation, therefore the absorbance goes up. Therefore, the path length is direct proportional to the concentration.
When the concentration is reported in moles/liter and the path length is reported in centimeters, the third factor is known equally the tooth absorptivity (\(\varepsilon\)). In some fields of piece of work, it is more mutual to refer to this every bit the extinction coefficient. When we use a spectroscopic method to measure the concentration of a sample, nosotros select out a specific wavelength of radiation to smooth on the sample. As yous probable know from other experiences, a particular chemical species absorbs some wavelengths of radiation and not others. The molar absorptivity is a measure of how well the species absorbs the detail wavelength of radiation that is being shined on it. The process of absorbance of electromagnetic radiation involves the excitation of a species from the ground country to a higher free energy excited state. This process is described every bit an excitation transition, and excitation transitions have probabilities of occurrences. It is advisable to talk about the degree to which possible free energy transitions within a chemical species are immune. Some transitions are more allowed, or more favorable, than others. Transitions that are highly favorable or highly allowed have loftier tooth absorptivities. Transitions that are simply slightly favorable or slightly allowed accept low tooth absorptivities. The higher the molar absorptivity, the higher the absorbance. Therefore, the tooth absorptivity is straight proportional to the absorbance.
If we return to the experiment in which a spectrum (recording the absorbance as a function of wavelength) is recorded for a compound for the purpose of identification, the concentration and path length are abiding at every wavelength of the spectrum. The only departure is the molar absorptivities at the different wavelengths, so a spectrum represents a plot of the relative molar absorptivity of a species as a part of wavelength.
Since the concentration, path length and tooth absorptivity are all direct proportional to the absorbance, we tin write the following equation, which is known as the Beer-Lambert law (often referred to as Beer'southward Police), to show this human relationship.
\[\mathrm{A = \varepsilon bc} \]
Note that Beer'south Law is the equation for a straight line with a y-intercept of zero.
If yous wanted to measure the concentration of a particular species in a sample, describe the procedure you would use to do so.
Measuring the concentration of a species in a sample involves a multistep process.
1 important consideration is the wavelength of radiation to apply for the measurement. Remember that the college the tooth absorptivity, the higher the absorbance. What this besides means is that the higher the tooth absorptivity, the lower the concentration of species that yet gives a measurable absorbance value. Therefore, the wavelength that has the highest molar absorptivity (\(\lambda\)max) is commonly selected for the analysis because it will provide the everyman detection limits. If the species you lot are measuring is one that has been ordinarily studied, literature reports or standard analysis methods will provide the \(\lambda\)max value. If information technology is a new species with an unknown \(\lambda\)max value, and so information technology is easily measured by recording the spectrum of the species. The wavelength that has the highest absorbance in the spectrum is \(\lambda\)max.
The 2nd stride of the process is to generate a standard bend. The standard curve is generated by preparing a series of solutions (commonly iii-5) with known concentrations of the species being measured. Every standard curve is generated using a bare. The blank is some appropriate solution that is assumed to have an absorbance value of zero. It is used to zilch the spectrophotometer before measuring the absorbance of the standard and unknown solutions. The absorbance of each standard sample at \(\lambda\)max is measured and plotted as a function of concentration. The plot of the data should exist linear and should get through the origin as shown in the standard curve in Figure \(\PageIndex{2}\). If the plot is not linear or if the y-intercept deviates substantially from the origin, it indicates that the standards were improperly prepared, the samples deviate in some way from Beer's Law, or that in that location is an unknown interference in the sample that is complicating the measurements. Bold a linear standard bend is obtained, the equation that provides the all-time linear fit to the data is generated.

Annotation that the slope of the line of the standard curve in Effigy \(\PageIndex{2}\) is (\(\varepsilon\)b) in the Beer'due south Police equation. If the path length is known, the gradient of the line can and so exist used to calculate the tooth absorptivity.
The third step is to measure the absorbance in the sample with an unknown concentration. The absorbance of the sample is used with the equation for the standard curve to summate the concentration.
Suppose a minor amount of stray radiation (PSouth) always leaked into your instrument and made it to your detector. This stray radiation would add to your measurements of Po and P. Would this crusade any deviations to Beer'southward law? Explain.
The fashion to think nearly this question is to consider the expression we wrote earlier for the absorbance.
\[\mathrm{A = \log\left(\dfrac{P_o}{P}\correct)} \]
Since stray radiation always leaks in to the detector and presumably is a fixed or constant quantity, we tin rewrite the expression for the absorbance including terms for the stray radiation. It is of import to recognize that Po, the power from the radiation source, is considerably larger than \(P_S\). Also, the numerator (Po + Ps) is a constant at a particular wavelength.
\[\mathrm{A = \log\left(\dfrac{P_o + P_s}{P + P_s}\right)} \]
At present let'southward examine what happens to this expression under the two extremes of low concentration and high concentration. At low concentration, non much of the radiations is absorbed and P is not that much different than Po. Since \(P_o \gg P_S\), \(P\) will besides be much greater than \(P_S\). If the sample is now made a little more concentrated so that a picayune more of the radiation is absorbed, P is still much greater than PS. Under these conditions the corporeality of stray radiation is a negligible contribution to the measurements of Po and P and has a negligible outcome on the linearity of Beer's Law.
As the concentration is raised, P, the radiation reaching the detector, becomes smaller. If the concentration is fabricated high enough, much of the incident radiation is absorbed past the sample and P becomes much smaller. If we consider the denominator (P + PS) at increasing concentrations, P gets modest and PS remains constant. At its limit, the denominator approaches PS, a constant. Since Po + PSouth is a abiding and the denominator approaches a constant (Ps), the absorbance approaches a constant. A plot of what would occur is shown in Figure \(\PageIndex{3}\).

The ideal plot is the direct line. The curvature that occurs at higher concentrations that is acquired by the presence of stray radiation represents a negative departure from Beer's Police force.
The derivation of Beer's Law assumes that the molecules absorbing radiations don't interact with each other (remember that these molecules are dissolved in a solvent). If the analyte molecules interact with each other, they can alter their power to blot the radiation. Where would this assumption break down? Guess what this does to Beer's law?
The sample molecules are more probable to collaborate with each other at higher concentrations, thus the assumption used to derive Beer'southward Police breaks down at high concentrations. The effect, which nosotros will not explain in any more detail in this document, also leads to a negative difference from Beer'south Constabulary at high concentration.
Beer'due south law also assumes purely monochromatic radiation. Describe an instrumental set upwards that would allow yous to polish monochromatic radiation on your sample. Is it possible to go purely monochromatic radiation using your gear up? Guess what this does to Beer'south law.
Spectroscopic instruments typically have a device known equally a monochromator. In that location are two key features of a monochromator. The first is a device to disperse the radiation into distinct wavelengths. You are likely familiar with the dispersion of radiation that occurs when radiation of unlike wavelengths is passed through a prism. The second is a slit that blocks the wavelengths that you do not want to smooth on your sample and only allows \(\lambda\)max to pass through to your sample every bit shown in Figure \(\PageIndex{4}\).
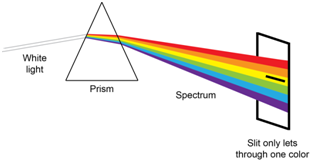
An examination of Effigy \(\PageIndex{4}\) shows that the slit has to permit some "packet" of wavelengths through to the sample. The packet is centered on \(\lambda\)max, only clearly nearby wavelengths of radiation laissez passer through the slit to the sample. The term constructive bandwidth defines the packet of wavelengths and it depends on the slit width and the ability of the dispersing element to dissever the wavelengths. Reducing the width of the slit reduces the packet of wavelengths that brand information technology through to the sample, meaning that smaller slit widths lead to more monochromatic radiation and less deviation from linearity from Beer's Law.
Is there a disadvantage to reducing the slit width?
The important thing to consider is the effect that this has on the power of radiation making it through to the sample (Po). Reducing the slit width will lead to a reduction in Po and hence P. An electronic measuring device called a detector is used to monitor the magnitude of Po and P. All electronic devices have a background noise associated with them (rather analogous to the static noise you may hear on a speaker and to the discussion of stray radiation from earlier that represents a form of dissonance). Po and P correspond measurements of indicate over the background noise. As Po and P become smaller, the background dissonance becomes a more significant contribution to the overall measurement. Ultimately the groundwork dissonance restricts the signal that can be measured and detection limit of the spectrophotometer. Therefore, information technology is desirable to have a big value of Po. Since reducing the slit width reduces the value of Po, information technology also reduces the detection limit of the device. Selecting the appropriate slit width for a spectrophotometer is therefore a rest or tradeoff of the desire for high source ability and the desire for high monochromaticity of the radiation.
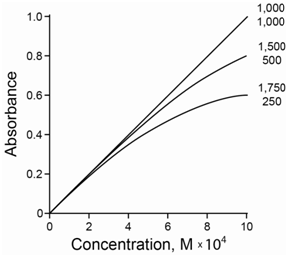
Information technology is non possible to get purely monochromatic radiation using a dispersing element with a slit. Normally the sample has a slightly different molar absorptivity for each wavelength of radiation shining on information technology. The net outcome is that the full absorbance added over all the different wavelengths is no longer linear with concentration. Instead a negative deviation occurs at higher concentrations due to the polychromicity of the radiation. Furthermore, the difference is more pronounced the greater the difference in the molar absorbtivity. Figure \(\PageIndex{v}\) compares the deviation for ii wavelengths of radiations with molar absorptivities that are (a) both 1,000, (b) 500 and 1,500, and (c) 250 and 1,750. As the tooth absorptivities become further apart, a greater negative deviation is observed.
Therefore, information technology is preferable to perform the absorbance measurement in a region of the spectrum that is relatively broad and flat. The hypothetical spectrum in Figure \(\PageIndex{vi}\) shows a species with two wavelengths that have the aforementioned tooth absorptivity. The peak at approximately 250 nm is quite sharp whereas the one at 330 nm is rather broad. Given such a pick, the broader elevation will take less divergence from the polychromaticity of the radiation and is less prone to errors caused by slight misadjustments of the monochromator.

Consider the relative error that would be observed for a sample as a function of the transmittance or absorbance. Is at that place a preferable region in which to measure the absorbance? What do yous recall about measuring absorbance values above 1?
It is of import to consider the error that occurs at the two extremes (loftier concentration and low concentration). Our discussion above virtually deviations to Beer'south Law showed that several problems ensued at higher concentrations of the sample. As well, the bespeak where only 10% of the radiation is transmitted through the sample corresponds to an absorbance value of 1. Because of the logarithmic relationship between absorbance and transmittance, the absorbance values rise rather rapidly over the concluding 10% of the radiation that is absorbed by the sample. A relatively small modify in the transmittance tin lead to a rather large alter in the absorbance at loftier concentrations. Because of the substantial negative deviation to Beer's law and the lack of precision in measuring absorbance values above i, it is reasonable to assume that the error in the measurement of absorbance would be high at loftier concentrations.
At very low sample concentrations, we observe that Po and P are quite similar in magnitude. If we lower the concentration a bit more, P becomes fifty-fifty more than similar to Po. The important realization is that, at low concentrations, we are measuring a pocket-size difference between two large numbers. For example, suppose we wanted to measure the weight of a captain of an oil tanker. One way to do this is to measure the combined weight of the tanker and the captain, so have the helm leave the ship and measure the weight once more. The deviation between these two large numbers would be the weight of the captain. If nosotros had a scale that was accurate to many, many significant figures, and then nosotros could perchance perform the measurement in this manner. But you likely realize that this is an impractical manner to accurately mensurate the weight of the captain and nearly scales practise not have sufficient precision for an accurate measurement. Similarly, trying to measure a small deviation betwixt two large signals of radiation is decumbent to error since the difference in the signals might be on the gild of the inherent racket in the measurement. Therefore, the degree of error is expected to be high at depression concentrations.
The word to a higher place suggests that it is best to measure the absorbance somewhere in the range of 0.1 to 0.8. Solutions of college and lower concentrations have higher relative fault in the measurement. Depression absorbance values (high transmittance) represent to dilute solutions. Often, other than taking steps to concentrate the sample, we are forced to measure samples that take depression concentrations and must accept the increased error in the measurement. It is generally undesirable to record absorbance measurements above 1 for samples. Instead, information technology is improve to dilute such samples and record a value that will be more precise with less relative mistake.
Another question that arises is whether it is acceptable to utilise a non-linear standard curve. As we observed earlier, standard curves of absorbance versus concentration volition evidence a non-linearity at higher concentrations. Such a not-linear plot can usually exist fit using a higher order equation and the equation may predict the shape of the curve quite accurately. Whether or not it is adequate to use the non-linear portion of the bend depends in part on the absorbance value where the non-linearity starts to appear. If the non-linearity occurs at absorbance values higher than one, it is usually better to dilute the sample into the linear portion of the curve because the absorbance value has a high relative error. If the non-linearity occurs at absorbance values lower than one, using a non-linear higher society equation to calculate the concentration of the analyte in the unknown may exist acceptable.
One matter that should never exist done is to extrapolate a standard curve to higher concentrations. Since non-linearity will occur at some betoken, and there is no way of knowing in advance when it will occur, the absorbance of any unknown sample must exist lower than the absorbance of the highest concentration standard used in the preparation of the standard bend. Information technology is also non desirable to extrapolate a standard curve to lower concentrations. In that location are occasions when not-linear furnishings occur at low concentrations. If an unknown has an absorbance that is below that of the lowest concentration standard of the standard bend, information technology is preferable to set up a lower concentration standard to ensure that the curve is linear over such a concentration region.
Another business organisation that ever exists when using spectroscopic measurements for chemical compound quantification or identification is the potential presence of matrix effects. The matrix is everything else that is in the sample except for the species beingness analyzed. A concern can occur when the matrix of the unknown sample has components in it that are not in the blank solution and standards. Components of the matrix tin have several undesirable effects.
What are some examples of matrix effects and what undesirable issue could each have that would compromise the absorbance measurement for a sample with an unknown concentration?
1 concern is that a component of the matrix may absorb radiation at the same wavelength as the analyte, giving a false positive signal. Particulate matter in a sample volition besprinkle the radiations, thereby reducing the intensity of the radiation at the detector. Scattered radiation will be confused with absorbed radiations and result in a higher concentration than really occurs in the sample.
Another business organisation is that some species have the power to modify the value of \(\lambda\)max. For some species, the value of \(\lambda\)max can show a pronounced dependence on pH. If this is a consideration, and then all of the standard and unknown solutions must be appropriately buffered. Species that can hydrogen bond or metal ions that tin can course donor-acceptor complexes with the analyte may change the position of \(\lambda\)max. Changes in the solvent can affect \(\lambda\)max besides.
burnettsaimandeper.blogspot.com
Source: https://chem.libretexts.org/Bookshelves/Analytical_Chemistry/Molecular_and_Atomic_Spectroscopy_(Wenzel)/1%3A_General_Background_on_Molecular_Spectroscopy/1.2%3A_Beers_Law
0 Response to "Do You Have to Start All Over Again if You Forgot to Zero Your Blank for Your Absorbances"
Post a Comment